 The other trend that can be observed is that, as you move down a group in the periodic table, the lattice energy decreases. For example, we can find the lattice energy of CaO \text 3430 kJ / mol. This kind of construction is known as a Born-Haber cycle. If we then add together all of the various enthalpies (if you don't remember the concept, visit our enthalpy calculator), the result must be the energy gap between the lattice and the ions. So, how to calculate lattice energy experimentally, then? The trick is to chart a path through the different states of the compound and its constituent elements, starting at the lattice and ending at the gaseous ions. These additional reactions change the total energy in the system, making finding what is the lattice energy directly difficult. This is because ions are generally unstable, and so when they inevitably collide as they diffuse (which will happen quite a lot considering there are over 600 sextillion atoms in just one mole of substance - as you can discover with our Avogadro's number calculator) they are going to react to form more stable products. While you will end up with all of the lattice's constituent atoms in a gaseous state, they are unlikely to still be in the same form as they were in the lattice. After this, the amount of energy you put in should be the lattice energy, right? 
Experimental methods and the Born-Haber cycleĪs one might expect, the best way of finding the energy of a lattice is to take an amount of the substance, seal it in an insulated vessel (to prevent energy exchange with the surroundings), and then heat the vessel until all of the substance is gas. 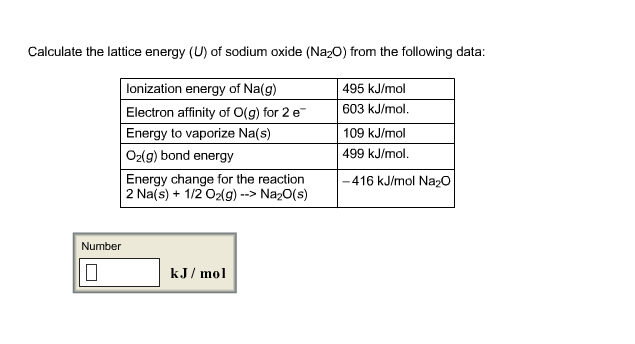
You can calculate the last four using this lattice energy calculator. We will discuss one briefly, and we will explain the remaining four, which are all slight variations on each other, in more detail. Perhaps surprisingly, there are several ways of finding the lattice energy of a compound. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
